
get a quote
Using an innovative optical geometry and standard x-ray tube, this instrument provides synchrotron-quality x-ray emission spectra (XES) at synchrotron-level count rates.
The ~1-eV energy resolution strongly outperforms all traditional WD-XRF systems in this energy range, with high-profile applications for sulfur and phosphorus speciation.
Tender X-Ray Spectrometers
easyXAFS
The BRIMSTONE is a high performance tender x-ray emission spectrometer (2-5 keV).Using an innovative optical geometry and standard x-ray tube, this instrument provides synchrotron-quality x-ray emission spectra (XES) at synchrotron-level count rates.
The ~1-eV energy resolution strongly outperforms all traditional WD-XRF systems in this energy range, with high-profile applications for sulfur and phosphorus speciation.
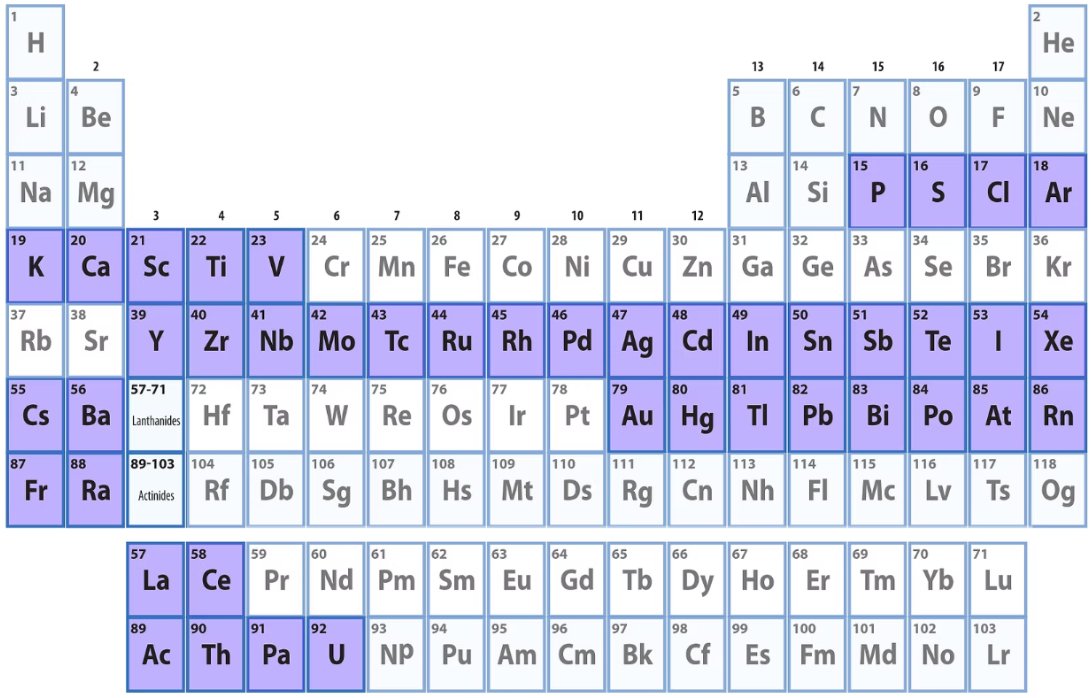
Tender X-ray instruments cover all the highlighted elements
Specifications
Rowland Circle Size: | 10 centimeters |
Measurement Mode: | XES only |
X-ray Source (XES): | 100-W air-cooled X-ray Tube |
Energy Range*: | Recommended: 2-5 keV |
| *Depends on selection of crystal analyzers | |
Data
Sulfur XES Kα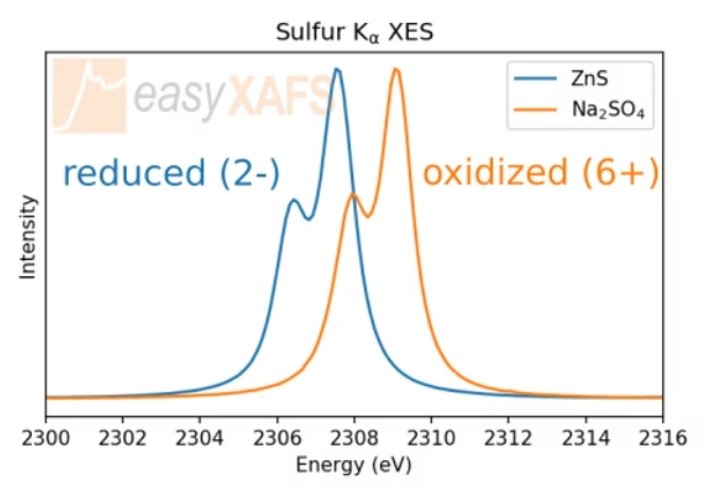 Tender X-ray Emission Spectroscopy (XES) measurements of the Sulfur Kα emission line in ZnS and Na2SO4 compounds. The change in oxidation state of the sulfur is indicated by the shift in energy of the spectrum, while there is very little change in spectral shape. |
Phosphorus XES Kα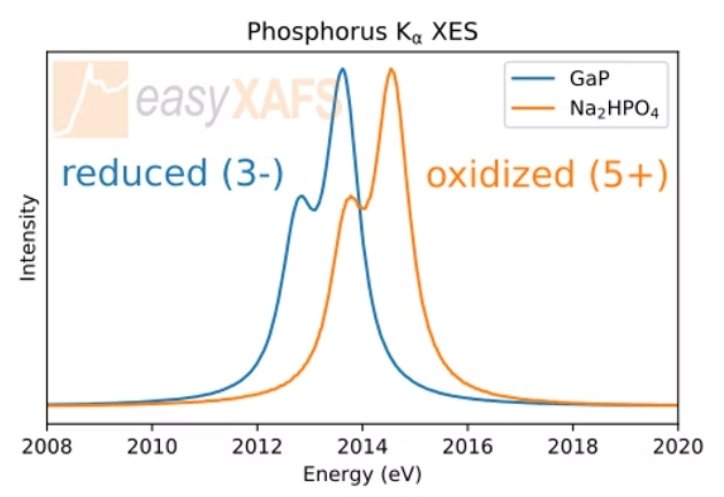 Tender XES measurements of the Phosphorus Kα emission line in GaP and Na2HPO4 compounds. The change in oxidation state of the phosphorus is indicated by the shift in energy of the spectrum, while there is very little change in spectral shape. |
Sulfur VTC-XES Kβ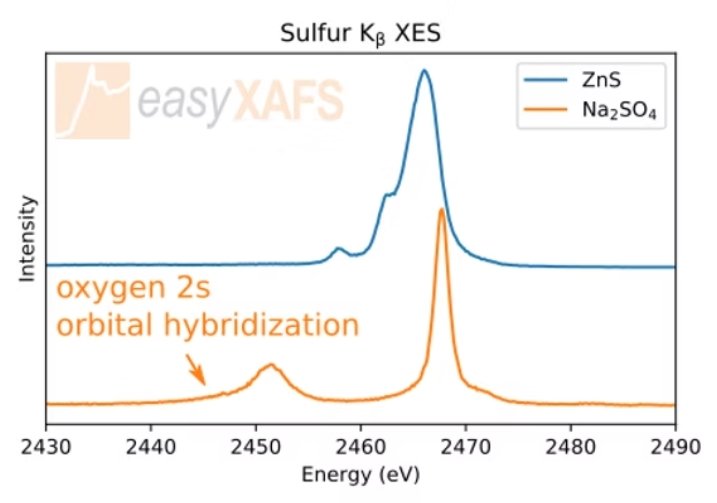 Tender XES measurements of Sulfur valence-to-core Kβ XES show an increased sensitivity to the valence electronic structure compared to the Kα. Comparing the measurements of ZnS and Na2SO4, a number of spectral features change, including the presence of a separate peak at lower energies due to the oxygen 2s orbital hybridization with the sulfur valence electrons. |
Phosphorus VTC-XES Kβ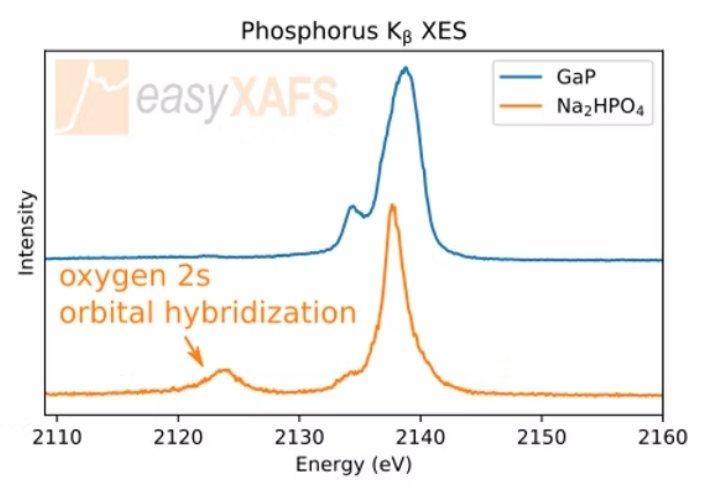 Tender XES measurements of Phosphorus valence-to-core Kβ XES show an increased sensitivity to the valence electronic structure compared to the Kα. Comparing the measurements of GaP and Na2HPO4, a number of spectral features change, including the presence of a separate peak at lower energies due to the oxygen 2s orbital hybridization with the phosphorus valence electrons. |
Sulfur Kalpha linear combination fitting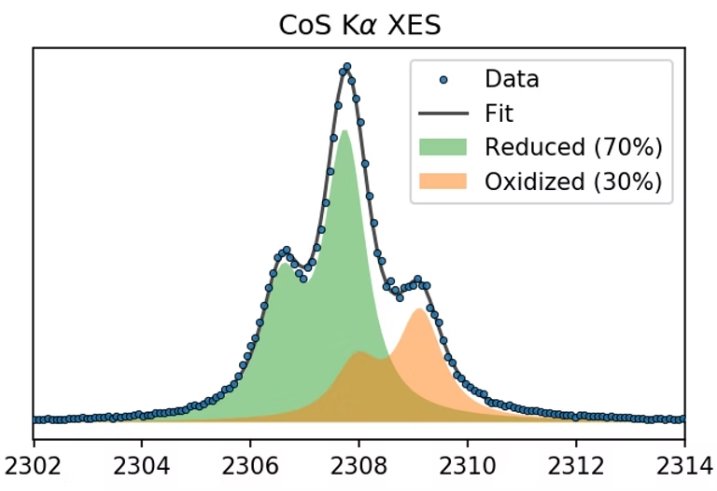 Sulfur Kα XES spectrum of a partially oxidized sample of CoS demonstrates the sensitivity of S Kα XES. Since the spectral shape does not change significantly, linear combination fitting can be performed with high confidence to determine the fraction of oxidized sulfur. |











